Dinçer UysalI; Fatih AksoyII; Erdogan IbrişimI
DOI: 10.21470/1678-9741-2019-0274
ABSTRACT
Objective: The aim of this study was to evaluate the value of CHA2DS2-VASc and Anticoagulation and Risk Factors in Atrial Fibrillation (ATRIA) risk scores for prediction of postoperative atrial fibrillation (AF) development in patients undergoing coronary artery bypass grafting (CABG) operation.AF = Atrial fibrillation
ALT = Alanine aminotransferase
AST = Aspartate aminotransferase
ATRIA = Anticoagulation and Risk Factors in Atrial Fibrillation
AUC = Area under the curve
CABG = Coronary artery bypass grafting
CI = Confidence interval
CVA = Cerebrovascular accident
HDL = High-density lipoprotein
IVSD = Interventricular septum diameter
LDL = Low-density lipoprotein
LVEDD = Left ventricular end-diastolic diameter
LVESD = Left ventricular end-systolic diameter
OR = Odds ratio
POAF = Postoperative atrial fibrillation
PWD = Posterior wall diameter
ROC = Receiver operating characteristics
TIA = Transient ischemic attack
INTRODUCTION
Postoperative atrial fibrillation (POAF) is the most common type of arrhythmia after cardiac surgery with the incidence ranging from 10 to 65%, and its prevalence is even higher in elderly patients with heart failure and severe left ventricular impairment[1]. Patients with developing atrial fibrillation (AF) after cardiac surgery have higher risk for morbidity, such as cerebrovascular accidents, pulmonary edema, longer hospital stays, and mortality compared to those who do not develop AF[1]. Preoperative determination of patients who may develop POAF and taking necessary precautions will decrease the mortality and morbidity rates. Therefore, scoring systems should be developed to predict the development of POAF.
The CHA2DS2-VASc and Anticoagulation and Risk Factors in Atrial Fibrillation (ATRIA) risk scores are cheap and easy scoring systems that are used to predict the risk of thromboembolism in non-valvular AF patients[2,3]. Additionally, these scoring systems have been shown to accurately predict worse clinical outcomes in patients undergoing coronary artery bypass grafting (CABG) regardless of having AF[4]. The components of these scoring systems, such as advanced age, presence of hypertension, presence of diabetes mellitus, low ejection fraction, and female gender, have been associated with poor outcomes, including recurrent ischemic events after cardiac surgery[5].
In this study, we aimed to investigate the predictive value of different thromboembolic risk scores in AF for the development of POAF.
METHODS
Study Population
This study included patients who underwent isolated CABG at the Suleyman Demirel University, Education and Research Hospital, between June 2017 and March 2019. The study population was retrospectively and consecutively analyzed by using our database, which was collected as a part of routine clinical practice. The overall study population included 400 patients undergoing CABG. Exclusion criteria were hyperthyroidism, age < 18 years, prior cardiac surgery, class III or IV heart failure, previous AF, left atrial diameter > 55 mm, left ventricular ejection fraction < 0.25, sepsis, heart rate < 60 bpm, systolic blood pressure < 90 mm Hg, inflammatory disease, and being already on antiarrhythmic treatment. According to these criteria, 30 patients were excluded due to previous AF (n=10), heart rate < 60 bpm (n=15), and hyperthyroidism (n=5). Therefore, 370 patients were included in this substudy. Informed consent was obtained from each patient, and the study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution's human research committee (Date: 28.05.2019, Decision no: 181). Similar operative techniques were used in all of the patients.
Diagnosis of Thromboembolic Risk
The CHA2DS2-VASc risk score is calculated by assigning a score of 1 point for each of the following conditions: congestive heart failure (ejection fraction < 40%), hypertension, age between 65 and 74 years, diabetes mellitus, vascular disease (myocardial infarction or peripheral arterial disease), and female gender; and a score of 2 points for the following conditions: history of stroke or transient ischemic attack (TIA) and age > 75 years. The score is then used to predict the risk of thromboembolism in non-valvular AF patients[2]. The maximum value of the score is 9.
The ATRIA score was developed from the ATRIA study cohort and calculated using the following: anemia (hemoglobin < 13 g/dL in men and < 12 g/dL in women) (3 points), severe renal disease (estimated glomerular filtration rate < 30 mL/min/1.73 m2) (3 points), age ≥ 75 years (2 points), prior bleeding, and hypertension. An ATRIA score of 0 to 3 is defined as “low risk”, a score of 4 is defined as “intermediate risk”, and a score ≥ 5 is defined as “high risk”[3]. The maximum value of the score is 12.
Rhythm Follow-up
The rhythms were followed by continuous electrocardiogram monitoring during intensive care unit stay and by all-day Holter during the rest of hospitalization. A 12-lead electrocardiogram was recorded every morning routinely and whenever the patients had symptoms suggestive of dysrhythmia. AF was defined as an irregular rhythm with the absence of discrete P waves in the 12-lead electrocardiogram. An AF episode lasting five minutes during hospitalization was defined as POAF[6]. In case of POAF, antiarrhythmics and electrical cardioversion were allowed based on the discretion of the physician.
Statistical Analysis
Statistical Package for the Social Sciences version 16.0 software package was used for statistical analyses in this study. Categorical variables were expressed as frequency (%) and compared using the χ2 test. Kolmogorov-Smirnov test was used to test the distribution of numeric variables; those with normal distribution were expressed as mean ± standard deviation and were compared with Student’s t-test. Data without normal distribution were expressed as median (interquartile range of 25%-75% percentiles) and were compared with the Mann-Whitney U test. In all statistical analyses, P-value < 0.05 was considered as statistically significant. The correlations between CHA2DS2-VASc and ATRIA risk scores, POAF, and other clinical, laboratory, and echocardiographic parameters were performed with Pearson’s or Spearman’s correlation analysis where appropriate. Univariate analysis and backward conditional binary logistic regression were performed to estimate the odds ratio (OR) and 95% confidence interval (CI) for the prediction of POAF. We carried out multivariate analysis in two models. Firstly, risk factors involved in CHA2DS2-VASc score were excluded from this analysis to avoid multicollinearity. Secondly, risk factors and other factors except CHA2DS2-VASc score were put to multivariate analysis. Receiver operating characteristics (ROC) curve analysis was used to analyze the prognostic value of CHA2DS2-VASc and ATRIA risk scores for POAF. C-statistic (area under the curve [AUC)] was presented as a unified estimate of sensitivity and specificity according to the cutoff value that was obtained by a ROC curve analysis. The optimal cutoff value was defined as the value yielding the maximal Youden index[7], or the best combined sensitivity and specificity. All ROC comparisons were performed using the DeLong test[8]. C-statistic (AUC) was presented as a unified estimate of sensitivity and specificity.
RESULTS
A total of 370 patients (mean age: 62.34±12 years; range, 28-84 years) were included in this study. During the follow-up period, 110 patients (29.7%) developed POAF. Demographic and clinical characteristics of the patients with and without POAF are listed in Table 1. Patients with POAF were significantly older and of the male gender when compared to patients without POAF (P<0.001 and P=0.03, respectively). Diabetes mellitus, hypertension, congestive heart failure, peripheral vascular disease, and stroke/transient ischemic event rates were higher in patients with POAF than in patients without POAF. There were no statistically significant differences between patients with and without POAF with regards to cholesterol parameters (for all parameters P>0.05). Left ventricular ejection fraction was significantly lower in patients with POAF than in patients without POAF (P<0.001). Preoperative fasting glucose levels were higher in patients with POAF than in patients without POAF (P<0.001).
| Without AF (n = 260) | With AF (n = 110) | P-value | |
|---|---|---|---|
| Age (years) | 59.8±12 | 68.1±8.4 | < 0.001 |
| Body mass index | 29.0±5.3 | 28.0±4.5 | 0.114 |
| Female gender (n, %) | 82 (31.5) | 24 (21.8) | 0.03 |
| Diabetes mellitus (n, %) | 108 (41.5) | 62 (56.4) | 0.006 |
| Hypertension (n, %) | 184 (70.8) | 100 (90.8) | < 0.001 |
| Congestive heart failure (n, %) | 12 (4.7) | 20. (18.9) | < 0.001 |
| Peripheral vascular disease (n, %) | 70(26.9) | 46(41.8) | 0.004 |
| History of CVA (n, %) | 36 (13.8) | 24 (21.8) | 0.04 |
| Ejection fraction (%) | 56.5±9.4 | 49.2±11.6 | < 0.001 |
| Left atrial diameter (mm) | 39.1±6.1 | 38.9±7.4 | 0.816 |
| LVEDD (mm) | 47.2±5.4 | 46.6±5.0 | 0.296 |
| LVESD (mm) | 29.6±6.8 | 31.3±5.7 | 0.026 |
| IVSD (mm) | 12.0±3.2 | 12.5±2.8 | 0.175 |
| PWD (mm) | 11.1±2.2 | 11.2±1.0 | 0.835 |
| Aortic diameter (mm) | 26.8±3.6 | 26.6±3.1 | 0.580 |
| Total cholesterol (mg/dl) | 201.2±41.3 | 196.9±39.4 | 0.412 |
| HDL cholesterol (mg/dl) | 41±10 | 42±9.8 | 0.713 |
| LDL cholesterol (mg/dl) | 126±40 | 125±34 | 0.915 |
| Triglycerides (mg/dl) | 160±106 | 142±49 | 0.209 |
| Creatinine (mg/dl) | 1.0±0.3 | 1.1±0.4 | 0.161 |
| ALT (U/L) | 22.2±12.2 | 27.5±29.8 | 0.015 |
| AST (U/L) | 31.1±23.6 | 33.0±22.7 | 0.489 |
| Glucose (mg/dl) | 142.5±67 | 198.6±119.9 | < 0.001 |
| CHA2DS2VASc score | 2.2±1.4 | 3.1±1.9 | < 0.001 |
| ATRIA risk score | 1.9±2.4 | 3.4±2.5 | < 0.001 |
Data presented as mean ± standard deviation or number (%) of the patients. CHA2DS2-VASc score stands for congestive heart failure, hypertension, age ≥ 75 years, diabetes mellitus, previous stroke, vascular disease, age 65 to 74 years, and female gender.
ALT=alanine aminotransferase; AST=aspartate aminotransferase; ATRIA=Anticoagulation and Risk Factors in Atrial Fibrillation; CVA=cerebrovascular accident; HDL=high-density lipoprotein; IVSD=interventricular septum diameter; LDL=low-density lipoprotein; LVEDD=left ventricular end-diastolic diameter; LVESD=left ventricular end-systolic diameter; PWD=posterior wall diameter
The postoperative drainage amounts in the first 24 hours and 48 hours were higher in patients with POAF than in patients without POAF (P=0.001 and P=0.05, respectively) (Table 2). Cardiopulmonary bypass time was longer in patients with POAF than in patients without POAF (P=0.02) but there is no statistically difference between patients with and without POAF with regards to clamp time. The duration of the hospitalization at the intensive care unit was longer in patients with POAF than in patients without POAF (P<0.001). There was no statistically difference between patients with and without POAF with regards to reoperation due to hemorrhage and intraoperative and in-hospital mortalities (for all parameters P>0.05) (Table 2).
| Without AF (n = 260) | With AF (n = 110) | P-value | |
|---|---|---|---|
| Cardiopulmonary bypass time (min) | 78.8±27.3 | 86.8±39.6 | 0.02 |
| Clamp time (min) | 47.6±15.4 | 47.0±16.4 | 0.766 |
| 24-hour drainage (ml) | 276.5±137 | 344.5±239 | 0.001 |
| 48-hour drainage (ml) | 145.3±82 | 162.7±71.1 | 0.05 |
| Duration of the hospitalization at the intensive care unit (days) | 2.1±0.4 | 2.6±1.4 | < 0.001 |
| Bypass number (n) | 2.4±2 | 2.4±0.7 | 0.806 |
| Reoperation due to hemorrhage (n, %) | 4 (1.5) | 2 (1.8) | 0.575 |
| Intraoperative mortality (n, %) | - | - | |
| In-hospital mortality (n, %) | 8 (3.1) | 4 (3.6) | 0.500 |
The mean CHA2DS2-VASc and ATRIA scores were significantly higher in patients with POAF than in patients without POAF (3.1±1.9 vs. 2.2±1.4, P<0.001; 3.4±2.5 vs. 1.9±2.4, P<0.001; respectively).
Prediction of Postoperative Atrial Fibrillation
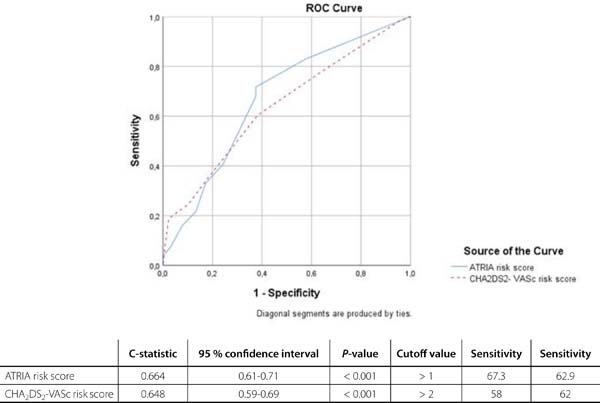
Univariate analyses showed that high CHA2DS2-VASc and ATRIA risk scores, diabetes mellitus, hypertension, congestive heart failure, peripheral vascular disease, stroke/TIA, low left ventricular ejection fraction, advanced age, fasting glucose level, serum alanine aminotransferase (ALT) level, 24-hour drainage amount, pumping time, and male gender were significantly associated with a higher risk of development of POAF (Table 3). Firstly, multivariate binary logistic regression analysis including characteristics - except for CHA2DS2-VASc and ATRIA scoring systems - associated with new-onset AF in univariate analysis showed that fasting glucose level (OR 1.009; 95% CI 1.006-1.013; P<0.001), age (OR 1.10; 95% CI 1.06-1.14; P<0.001), left ventricular ejection fraction (OR 0.91; 95% CI 0.89-0.94; P<0.001), male gender (OR 1.94; 95% CI 0.93-4.07; P=0.076), 24-hour drainage amount (OR 1.002; 95% CI 1.001-1.004; P=0.002), and serum ALT level (OR 1.022; 95% CI 1.001-1.043; P=0.038) remained independent factors for incident AF (Table 4). After that, multivariate binary logistic regression analysis including characteristics - except for diabetes mellitus, stroke/transient ischemic disease, peripheral vascular disease, male gender, congestive heart failure, hypertension, age, and left ventricular ejection fraction - associated with new-onset AF in univariate analysis showed that ATRIA risk score (OR 1.232; 95% CI 1.11-1.36; P<0.001), fasting glucose level (OR 1.006; 95% CI 1.004-1.009; P<0.001), and 24-hour drainage amount were associated with new-onset AF following CABG (OR 1.002; 95% CI 1.001-1.004; P<0.001) (Table 4). ROC curve analysis showed that both ATRIA score (C-statistic 0.66; 95% CI 0.61-0.71; P<0.001) and CHA2DS2-VASc score (C-statistic 0.64; 95% CI 0.59-0.69; P<0.001) were significant predictors of POAF (Figure 1). We calculated that a cutoff point of 1 for ATRIA and 2 for CHA2DS2-VASc scores could estimate the presence of POAF with a sensitivity of 67% and 62% and a specificity of 58% and 62%, respectively. We performed a pairwise comparison of ROC curves and recorded that the predictive value of ATRIA risk score with regards to POAF development was similar to that of CHA2DS2-VASc score (by DeLong method, AUCATRIAvs. AUCCHA2DS2-VASc z-test=0.568, P=0.569)
| Odds ratio | Confidence interval | P-value | |
|---|---|---|---|
| CHA2DS2-VASc risk score | 1.38 | 1.21-1.59 | < 0.001 |
| ATRIA risk score | 1.24 | 1.14-1.36 | < 0.001 |
| Fasting glucose level | 1.007 | 1.004-1.009 | < 0.001 |
| Age | 1.08 | 1.05-1.11 | < 0.001 |
| Male gender | 1.65 | 0.97-2.78 | 0.06 |
| ALT level | 1.014 | 1.00-1.02 | 0.03 |
| 24-hour drainage | 1.002 | 1.001-1.003 | 0.001 |
| 48-hour drainage | 1.003 | 1.000-1.005 | 0.056 |
| Left ventricular ejection fraction | 0.93 | 0.91-0.96 | < 0.001 |
| Cardiopulmonary bypass time | 1.008 | 1.001-1.015 | 0.03 |
| Model 1 | Model 2 | |||||
|---|---|---|---|---|---|---|
| Odds ratio | Confidence interval |
P-value | Odds ratio | Confidence interval |
P-value | |
| 24-hour drainage | 1.002 | 1.001-1.004 | 0.002 | 1.002 | 1.001-1.004 | < 0.001 |
| Age | 1.10 | 1.06-1.14 | < 0.001 | |||
| Male gender | 1.94 | 0.93-4.07 | 0.076 | |||
| Left ventricular ejection fraction | 0.91 | 0.89-0.94 | < 0.001 | |||
| ATRIA risk score | 1.232 | 1.11-1.36 | < 0.001 | |||
| Fasting glucose level | 1.009 | 1.006-1.013 | < 0.001 | 1.006 | 1.004-1.009 | < 0.001 |
| Serum ALT level | 1.022 | 1.001-1.043 | 0.038 | |||
Model 1: risk factors and other factors except CHA2DS2-VASc and ATRIA risk scores. Model 2: variables except risk factors involved in CHA2DS2-VASc and ATRIA risk scores.
CHA2DS2-VASc stands for congestive heart failure, hypertension, age ≥ 75 years, diabetes mellitus, previous stroke, vascular disease, age 65 to 74 years, and female gender
ALT=alanine aminotransferase; ATRIA=Anticoagulation and Risk Factors in Atrial Fibrillation
DISCUSSION
The current study showed that higher CHA2DS2-VASc and ATRIA scores were independently associated with the development of AF in patients undergoing CABG; consequently, both scores could be helpful and appropriate scoring systems for predicting AF after CABG.
Previous studies showed that hypertension, diabetes mellitus, obesity, valvular disease, increased age, and left atrial characteristics, such as size, volume, and scarring, contribute to the development of POAF[9]. Similarly, hypertension and diabetes mellitus rates were higher in patients with POAF than without POAF. The risk factors of POAF are similar to the components of CHA2DS2-VASc and ATRIA risk scores[1]. Furthermore, these scores can be used to predict the risk of POAF. Kashani et al.[10] showed that the CHA2DS2-VASc score can be used as a simple and useful tool for predicting POAF in patients with undergoing CABG. Borde et al.[11] carried out a study including 729 CABG patients and showed that higher CHA2DS2-VASc scores predict POAF. Parallel results were attained by Chua et al.[12], based on a prospective study including 277 patients undergoing CABG and/or valve procedures. Moreover, we showed that CHA2DS2-VASC score have been predicting AF following ST-elevation myocardial infarction and associated with epicardial fat tissue and mitral annular calcification[13-15]. The data of the current study corroborate with the results of previous studies. Additionally, we showed that the ATRIA score can also be used for the prediction of AF following CABG. Moreover, ATRIA score was similar with CHA2DS2-VASc score in predicting POAF.
POAF is a serious complication of CABG and is associated with worse clinical outcomes such as prolonged length of hospital stay, rising costs, and increased short- and long-term morbidity and mortality[16]. Several guidelines recommended suggestions for prophylaxis against POAF[17-19]. However, these suggestions reveal unnecessary exposure to medications and for this reason, many physicians do not regularly apply them. These propositions may expose up to 70% of undergoing CABG patients to antiarrhythmic drugs and their subsequent side effects[20]. In a randomized study, Ozaydın et al.[6] showed that carvedilol plus N-acetylcysteine decreased POAF incidence compared with metoprolol or carvedilol. Therefore, it is important to anticipate which patients may develop POAF. In this study, we demonstrated that CHA2DS2-VASc and ATRIA scoring systems could be used to determine patients at the highest risk of developing POAF, thus these scoring systems may decrease nonselective prophylaxis.
Additionally, the present study showed that older age, male gender, drainage amount, pumping time, and ALT levels were associated with POAF. The results corroborated with the previous studies[1,6,9,16,19,21]. Age is the most common risk factor of POAF because of occurred myocardial fiber loss, fibrosis, and collagen deposition[22]. Previous studies showed that cross-clamp duration and pumping time are associated with POAF, and reducing cross-clamp duration may reduce POAF[1,21,23].
CONCLUSION
In conclusion, we have shown in the current study that ATRIA and CHA2DS2-VASc scoring systems were useful for detecting AF following CABG. Additionally, when the ATRIA risk score was compared with the CHA2DS2-VASc scoring system, it was found to be powerful in predicting the development of POAF. Patients at high risk according to ATRIA and CHA2DS2-VASc scoring systems should be followed up for rhythm disturbance after the procedure and it should be administrated the suggestions of the guidelines prior to the procedure.
REFERENCES
1. Maisel WH, Rawn JD, Stevenson WG. Atrial fibrillation after cardiac surgery. Ann Intern Med. 2001;135(12):1061-73. doi:10.7326/0003-4819-135-12- 200112180-00010.
2. Kirchhof P, Benussi S, Kotecha D, Ahlsson A, Atar D, Casadei B, et al. 2016 ESC guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J. 2016;37(38):2893-962. doi:10.1093/ eurheartj/ehw210.
3. Singer DE, Chang Y, Borowsky LH, Fang MC, Pomernacki NK, Udaltsova N, et al. A new risk scheme to predict ischemic stroke and other thromboembolism in atrial fibrillation: the ATRIA study stroke risk score. J Am Heart Assoc. 2013;2(3):e000250. doi:10.1161/JAHA.113.000250.
4. Kalyoncuoglu M, Ozturk S, Sahin M. Does CHA2DS2-VASc score predict MACE in patients undergoing isolated coronary artery bypass grafting surgery? Braz J Cardiovasc Surg. 2019;34(5):542-9. doi:10.21470/1678-9741-2018-0323.
5. Kamal YA, Al-Elwany SE, Ghoneim AM, El-Minshawy AK. Predictors of early adverse events after isolated coronary artery bypass grafting: current status. Austin J Clin Cardiolog. 2017;4(2):1059.
6. Ozaydin M, Icli A, Yucel H, Akcay S, Peker O, Erdogan D, et al. Metoprolol vs. carvedilol or carvedilol plus n-acetyl cysteine on post-operative atrial fibrillation: a randomized, double-blind, placebo-controlled study. Eur Heart J. 2013;34(8):597-604. doi:10.1093/eurheartj/ehs423.
7. Youden WJ. Index for rating diagnostic tests. Cancer. 1950;3(1):32-5. doi:10.1002/1097-0142(1950)3:1<32::aid-cncr2820030106>3.0.co;2-3.
8. DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837-45. doi:10.2307/2531595.
9. Maesen B, Nijs J, Maessen J, Allessie M, Schotten U. Post-operative atrial fibrillation: a maze of mechanisms. Europace. 2012;14(2):159-74. doi:10.1093/ europace/eur208.
10. Kashani RG, Sareh S, Genovese B, Hershey C, Rezentes C, Shemin R, et al. Predicting postoperative atrial fibrillation using CHA2DS2-VASc scores. J Surg Res. 2015;198(2):267-72. doi:10.1016/j.jss.2015.04.047.
11. Borde D, Gandhe U, Hargave N, Pandey K, Mathew M, Joshi S. Prediction of postoperative atrial fibrillation after coronary artery bypass grafting surgery: is CHA 2 DS 2-VASc score useful? Ann Card Anaesth. 2014;17(3):182-7. doi:10.4103/0971-9784.135841.
12. Chua SK, Shyu KG, Lu MJ, Lien LM, Lin CH, Chao HH, et al. Clinical utility of CHADS2 and CHA2DS2-VASc scoring systems for predicting postoperative atrial fibrillation after cardiac surgery. J Thorac Cardiovasc Surg. 2013;146(4):919-26.e1. doi:10.1016/j.jtcvs.2013.03.040.
13. Aksoy F, Guler S, Kahraman F, Kuyumcu MS, Bagci A, Bas HA, et al. The relationship between mitral annular calcification, metabolic syndrome and thromboembolic risk. Braz J Cardiovasc Surg. 2019;34(5):535-41. doi:10.21470/1678-9741-2019-0062.
14. Aksoy F, Baş HA, Bağcı A, Oskay T. The CHA2DS2-VASc score for predicting atrial fibrillation in patients presenting with ST elevation myocardial infarction: prospective observational study. Sao Paulo Med J. 2019;137(3):248- 54. doi:10.1590/1516-3180.2018.0431140319.
15. Aksoy F, Guler S, Kahraman F, Oskay T, Varol E. The relation between echocardiographic epicardial fat thickness and CHA2DS2-VASc score in patients with sinus rhythm. Braz J Cardiovasc Surg. 2019;34(1):41-7. doi:10.21470/1678-9741-2018-0230.
16. Hernández-Leiva E, Alvarado P, Dennis RJ. Postoperative atrial fibrillation: evaluation of its economic impact on the costs of cardiac surgery. Braz J Cardiovasc Surg. 2019;34(2):179-86. doi:10.21470/1678-9741-2018-0218.
17. Fuster V, Rydén LE, Cannom DS, Crijns HJ, Curtis AB, Ellenbogen KA, et al. 2011 ACCF/AHA/HRS focused updates incorporated into the ACC/AHA/ ESC 2006 guidelines for the management of patients with atrial fibrillation: a report of the American college of cardiology foundation/American heart association task force on practice guidelines developed in partnership with the European society of cardiology and in collaboration with the European heart rhythm association and the heart rhythm society. J Am Coll Cardiol. 2011;57(11):e101-98. doi:10.1016/j.jacc.2010.09.013.
18. Bradley D, Creswell LL, Hogue Jr CW, Epstein AE, Prystowsky EN, Daoud EG. Pharmacologic prophylaxis: American college of chest physicians guidelines for the prevention and management of postoperative atrial fibrillation after cardiac surgery. Chest. 2005;128(2 Suppl):39S-47S. doi:10.1378/ chest.128.2_suppl.39s.
19. Fernando HC, Jaklitsch MT, Walsh GL, Tisdale JE, Bridges CD, Mitchell JD, et al. The society of thoracic surgeons practice guideline on the prophylaxis and management of atrial fibrillation associated with general thoracic surgery: executive summary. Ann Thorac Surg. 2011;92(3):1144-52. doi:10.1016/j. athoracsur.2011.06.104.
20. Mariscalco G, Biancari F, Zanobini M, Cottini M, Piffaretti G, Saccocci M, et al. Bedside tool for predicting the risk of postoperative atrial fibrillation after cardiac surgery: the POAF score. J Am Heart Assoc. 2014;3(2):e000752. doi:10.1161/JAHA.113.000752.
21. Mathew JP, Fontes ML, Tudor IC, Ramsay J, Duke P, Mazer CD, et al. A multicenter risk index for atrial fibrillation after cardiac surgery. JAMA. 2004;291(14):1720-9. doi:10.1001/jama.291.14.1720.
22. Greenberg JW, Lancaster TS, Schuessler RB, Melby SJ. Postoperative atrial fibrillation following cardiac surgery: a persistent complication. Eur J Cardiothorac Surg. 2017;52(4):665-72. doi:10.1093/ejcts/ezx039.
23. Mathew JP, Parks R, Savino JS, Friedman AS, Koch C, Mangano DT, et al. Atrial fibrillation following coronary artery bypass graft surgery: predictors, outcomes, and resource utilization. Multicenter study of perioperative ischemia research group. JAMA. 1996;276(4):300-6.
Authors' roles & responsibilities
DU Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work; final approval of the version to be published
FA Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work; final approval of the version to be published
EI Substantial contributions to the conception or design of the work; or the acquisition, final approval of the version to be published
Article receive on Friday, July 5, 2019
Article accepted on Monday, October 14, 2019
 All scientific articles published at www.rbccv.org.br are licensed under a Creative Commons license
All scientific articles published at www.rbccv.org.br are licensed under a Creative Commons license